

Structure classification Saturated hydrocarbons are having only single covalent bonds between their carbons.An group, generally abbreviated with the symbol R, is a functional group that, like an alkane, consists solely of single-bonded carbon and hydrogen atoms connected acyclically-for example, a. The alkanes have two main commercial sources: (crude oil) and.
They can be viewed as molecular trees upon which can be hung the more active/reactive of biological molecules. With their repeated -CH 2- units, the alkanes constitute a of organic compounds in which the members differ in by multiples of 14.03 (the total mass of each such unit, which comprises a single carbon atom of mass 12.01 u and two hydrogen atoms of mass ~1.01 u each).Īlkanes are not very reactive and have little. One group of the are, solids at standard and pressure (SATP), for which the number of carbons in the carbon backbone is greater than about 17. The number of carbon atoms may be thought of as the size of the alkane. The longest series of linked carbon atoms in a molecule is known as its or carbon backbone. In an alkane, each carbon atom has 4 bonds (either C-C or ), and each hydrogen atom is joined to one of the carbon atoms (so in a C-H bond). Besides this standard definition by, in some authors' usage the term alkane is applied to any saturated hydrocarbon, including those that are either monocyclic (i.e. The alkanes range in complexity from the simplest case of, CH 4 where n = 1 (sometimes called the parent molecule), to arbitrarily large molecules. Alkanes have the general chemical formula n 2 n+2. In other words, an alkane consists of and atoms arranged in a structure in which all the are.
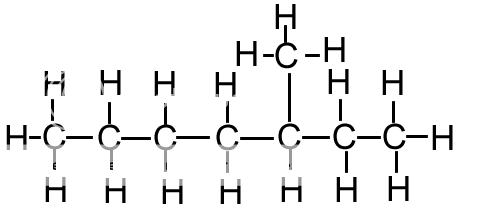
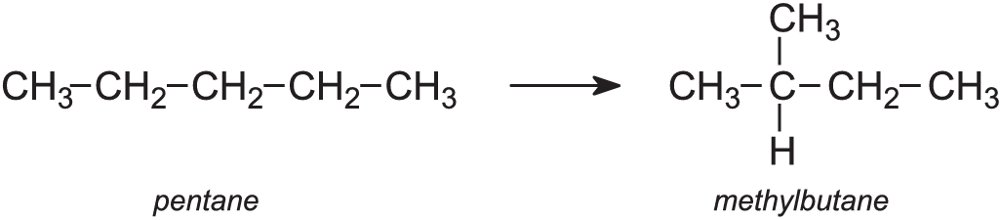
(ii) A molecule of decane contains ten carbon atoms.Ĭhemical structure of, the simplest alkane In, an alkane, or paraffin (a historical name that also has ), is an. Write a balanced equation for the cracking of nonane into heptane and ethene. A molecule of decane contains ten carbon.

 RSS Feed
RSS Feed
